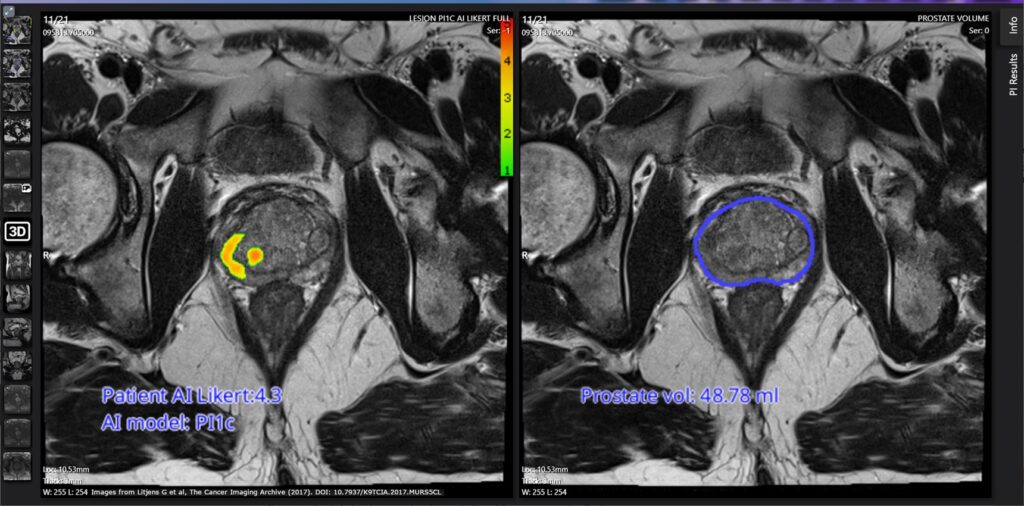
Cancer detection company Lucida Medical and leading doctors in Cambridge and Hampshire launch a £1million project to develop precision medicine technology to identify and manage the most common cancer in men.
CAMBRIDGE, UK (30 January 2024) – Lucida Medical, the University of Cambridge and Hampshire Hospitals NHS Foundation Trust have been awarded funding by Innovate UK and the Office for Life Sciences to extend the ground-breaking Pi software platform to multiple clinical indications. Already certified to support cancer diagnosis, this £1million programme will enable Pi to be used throughout a patient’s journey with prostate cancer, enabling more accurate and less invasive testing by using artificial intelligence (AI) to combine insights from MRI and clinical tests
More than 12,000 die each year
Over 50,000 men each year are diagnosed with prostate cancer in the UK each year, and 490,000 live with or have been treated for the disease. 1.4 million men are diagnosed annually worldwide. While prostate cancer may initially develop slowly, it can be deadly if found too late, killing more than 12,000 in the UK and 375,000 worldwide each year.
Project aims to offer active surveillance
This project will extend the Pi platform to further support active surveillance of patients living with prostate cancer. Dr Tristan Barrett, Associate Professor of Radiology at the University of Cambridge, said: “In our clinical work, we offer active surveillance to patients with lower risk prostate cancer, with regular imaging and blood tests to track the cancer. The aim is to delay surgery or other therapies that can have serious side effects. We have shown this to be a safe approach, which enables most men to retain their quality of life for many years.”
Dr Aarti Shah, Consultant Radiologist at Hampshire Hospitals NHS Foundation Trust, added: “Monitoring patients on active surveillance involves regular MRI scans, blood tests and occasionally biopsies. The analysis of multiple MRI scans is a time-consuming task for expert radiologists, of whom there are too few in the UK. We are excited to be able to develop cutting-edge tools to offer patients and clinical teams a smarter approach to active surveillance through this project.”
Research to predict and track cancer progress
Research by Lucida Medical and by the University of Cambridge has demonstrated how to predict whether prostate cancer is significant and track its evolution, more accurately by using AI methods. In this programme, Hampshire Hospitals NHS Foundation Trust will lead the development of a new dataset of patients on active surveillance. Lucida Medical will use this to train new AI algorithms and develop additional workflows to extend the Pi platform. The University of Cambridge will then evaluate the Pi platform with data from prostate cancer patients dating back over the last 10 years.
The programme costs are covered by Lucida Medical with support from Innovate UK’s Advancing Precision Medicine programme, including funding from the Office of Life Sciences.
“With this collaboration, Pi will enable us to test and monitor cancer patients using precision medicine across the whole prostate cancer pathway. This will reduce the impact of the disease for both patients and hospitals and help ensure patients receive treatment exactly when they need it,” commented Dr Antony Rix, CEO and co-founder of Lucida Medical.
Lucida Medical will demonstrate Pi at the 2024 European Congress of Radiology, where Dr Rix will present the results of the latest research on the Pi platform in session RPS 605 on 28 February from 16:30.
Pi is available for use in the UK and EU to support the diagnosis of prostate cancer.